
If the solute dissociates into 2 ions in solution, then 1 mole of the solute will contain 2 Osm. A solution containing either 1 mole of glucose (180 g) or 1 mole of albumin (70,000 g) in 1 kg of water has a concentration of 1 Osm/kg of water, because neither glucose nor albumin dissociates in solution. 
One mole of a pure substance has a mass in grams equal to its molecular weight. 
One osmole is equal to 1 mole of an undissociated solute. Because it is the number and not the size or type of solute particles that causes water movement and hence contributes to the osmotic pressure of a solution, the term osmole (Osm, or osmol) is used to describe the number of osmotically active solute particles, regardless of their mass. Therefore it is useful to have a concentration term that refers to the total concentration of solute particles that causes osmotic movement of water. Therefore at equilibrium the osmolar concentration (osmolarity) of the intracellular and interstitial fluid compartments remain similar, at approximately 290 mOsm/L.Īs noted, a difference in the solute concentrations of two fluid compartments separated by a semipermeable membrane causes osmotic movement of water. When this happens, net influx of water causes the cell to expand, whereas net efflux of water causes the cell to contract. However, under certain conditions, when a concentration difference for water develops across the cell membrane by active transport of solutes, osmotic forces will develop across the cell membrane and water will move rapidly between these two compartments until an osmotic equilibrium is achieved. In the steady state, the volume of water that diffuses across the membrane in either direction is balanced precisely so that no net diffusion of water occurs and the volume of the cell remains unchanged. In some cells, the number of AQPs, and thus the permeability to water, can be altered in response to hormones. The number of AQPs, also known as water channels, differs in membranes of different tissues. Although water is a polar molecule, it is able to penetrate the nonpolar lipid region of membranes through a group of transmembrane channel proteins called aquaporins (AQPs), which form channels through which water can readily diffuse. Most cell membranes are semipermeable, that is, relatively impermeable to most solutes but highly permeable to water. The process of osmosis also explains the movement of water across cell membranes. This pressure is called the osmotic pressure. As shown in diagram III, application of a pressure can prevent osmotic movement of water across the semipermeable membrane. Because the volume of compartment B increased, there were no significant changes in hydrostatic pressure in the compartments. In this example, the volume of compartment B increased, whereas the volume of compartment A decreased. The movement of water across the semipermeable membrane leads to a change in the initial volumes at equilibrium. There is net movement of water from A to B until the solutions in the two compartments are iso-osmotic, as shown in diagram II. In diagram I, compartments A and B are shown filled with equal volumes of solution, but the solution in compartment A is hypo-osmotic with reference to the solution in compartment B. 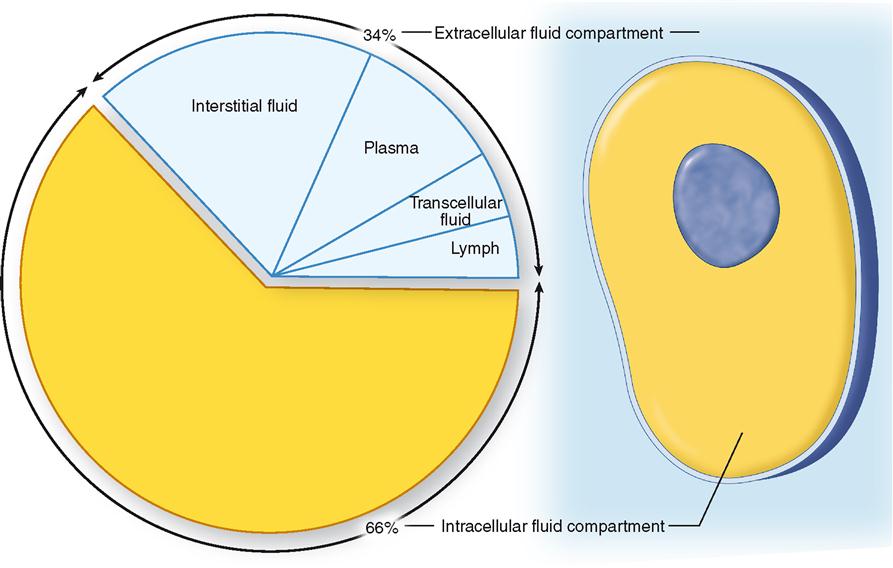
This is achieved by removing excess heat from the body by evaporative water loss from the skin.įIGURE 35-2 Osmosis and osmotic pressure can be illustrated by two compartments separated by a semipermeable membrane, permeable to water but not to solutes (circles). Another important physiological function of water is its role in the regulation of body temperature. The intracellular water establishes the physicochemical medium that allows various metabolic processes to take place. The extracellular water bathing the cells serves as a medium for the transport of nutrients and oxygen to the cells and for removing wastes from the cells, which will be eliminated by the liver and kidneys. It is the principal fluid medium in which nutrients, minerals, gases, and enzymes are dissolved. Water performs several functions that are essential to life. The volume of the intracellular fluid provides turgor to the tissues, which is important for the tissue or organ form and ultimately the body form. It lubricates joints and moistens tissues such as those in the eyes, nose, and mouth. In the body, water is present inside and around the cells and within all blood vessels. Water is an essential nutrient vital to the existence of both animals and plants.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
